PT-141 (Bremelanotide)
Desire Starts in the Brain. So Does This.
What It Treats

How It Works
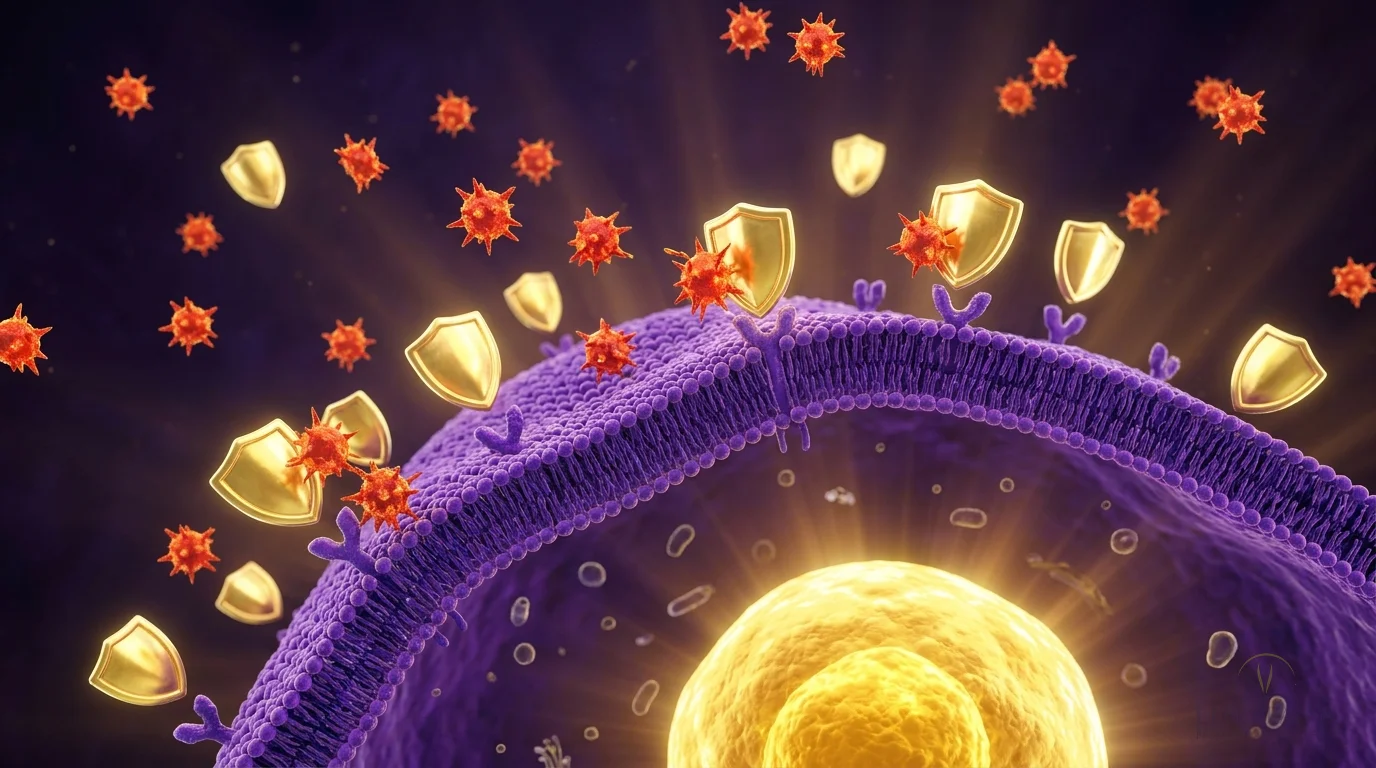
PT-141 activates melanocortin receptors (MC3R/MC4R) in the hypothalamus — the brain's desire center. Unlike Viagra (which works on blood vessels), PT-141 enhances actual desire and arousal at the neurological level.
Mechanism of Action
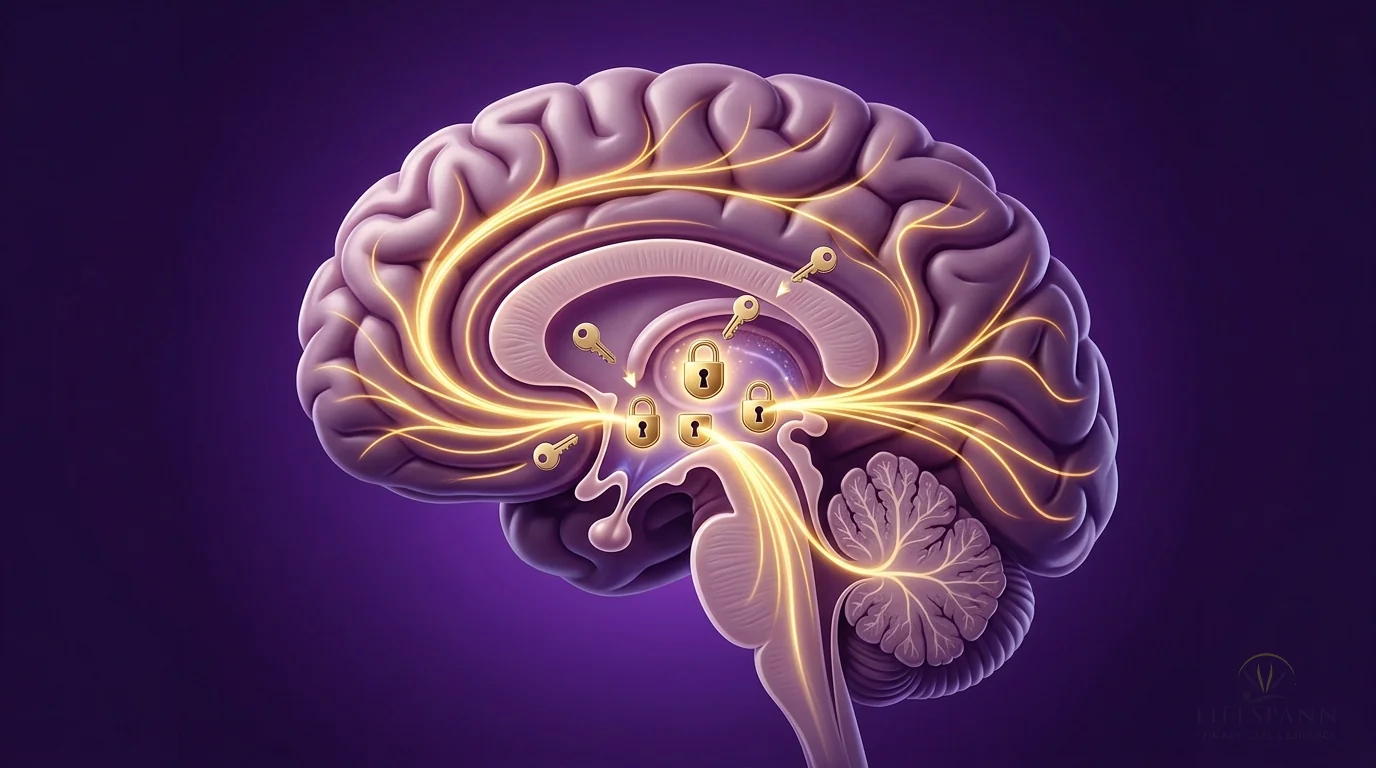
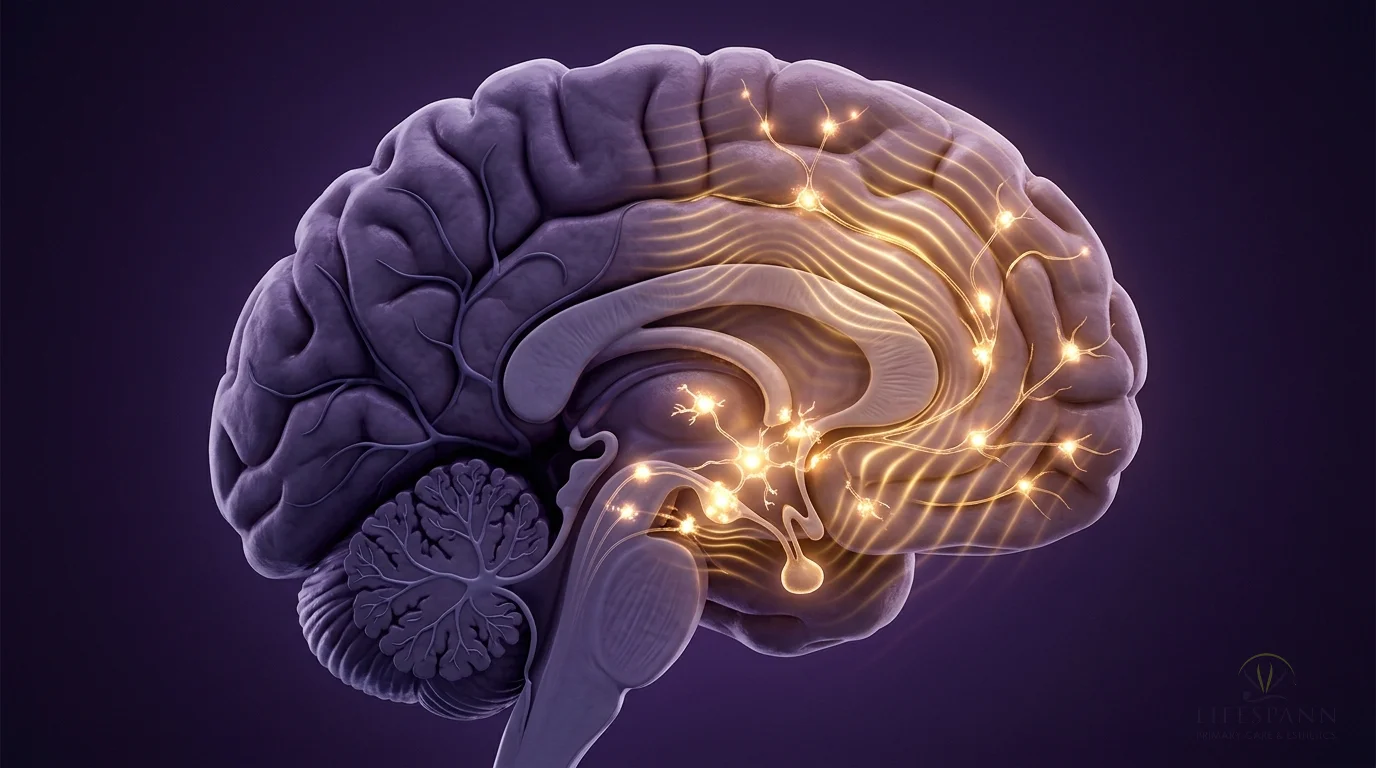
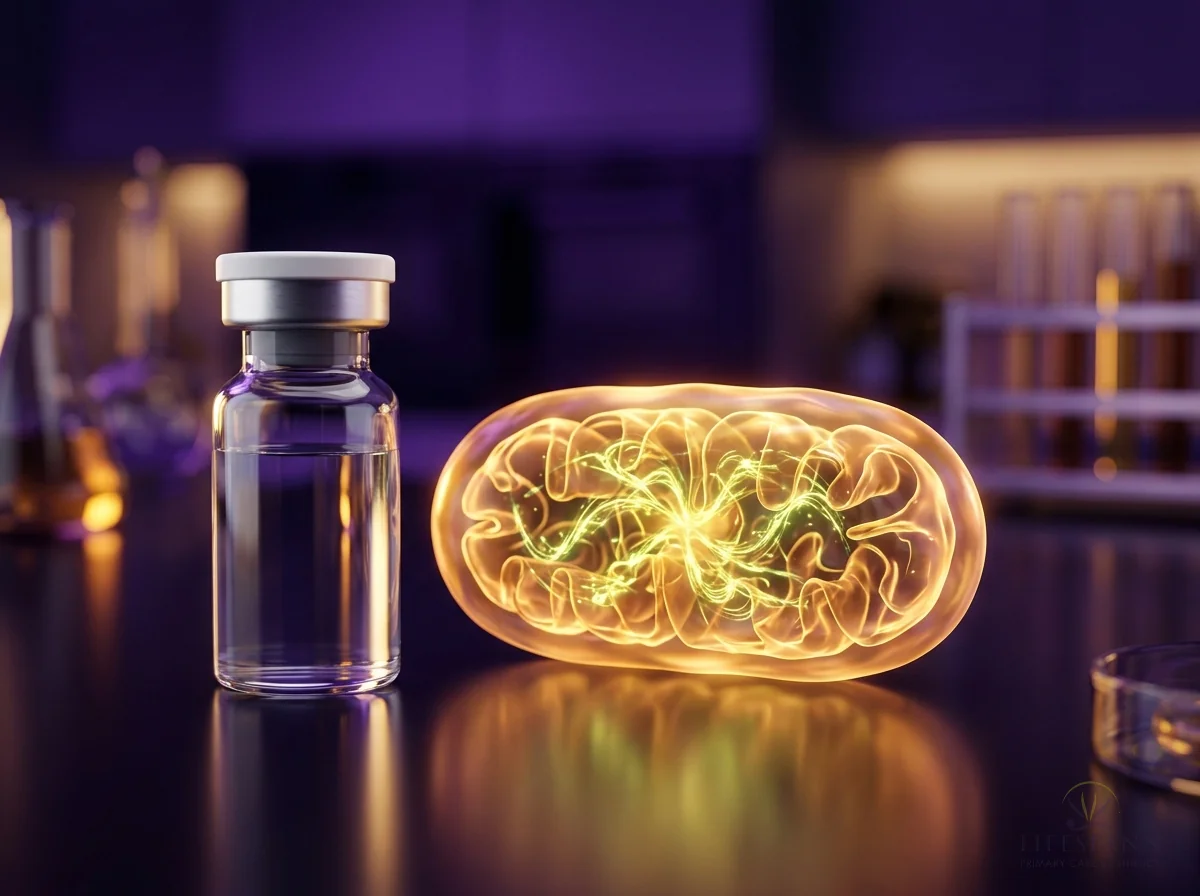
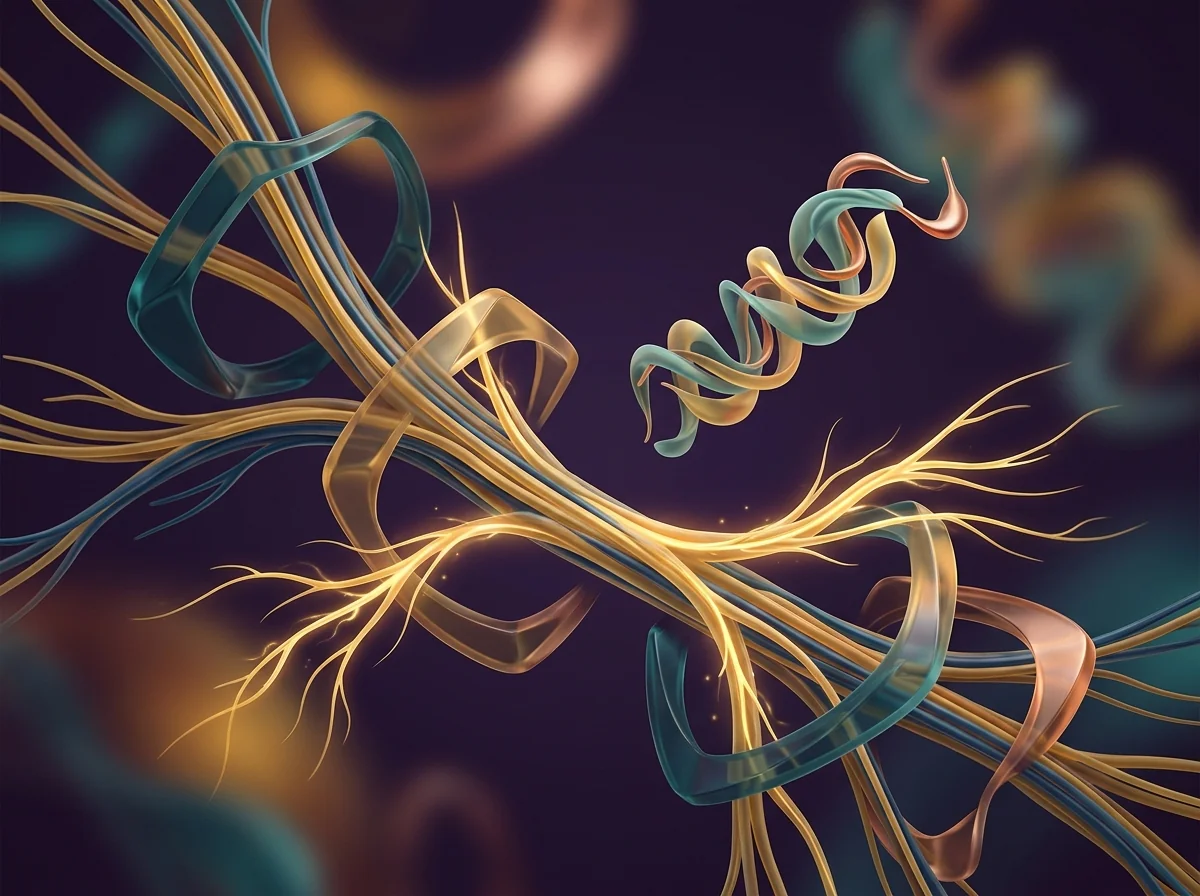
Here's why PT-141 is different from anything else on the market, and why we recommend it to both men and women. Viagra and similar medications work on blood flow — they're plumbing fixes. PT-141 works in the brain, where desire actually starts. In this illustration, you can see golden pathways lighting up deep inside the brain's hypothalamus. A 2021 review in the World Journal of Men's Health found that in a trial of 342 men who didn't respond to Viagra, PT-141 produced successful erectile response in 34% versus just 9% on placebo — because it works through a completely separate pathway in the brain. The golden lock-and-key symbols show PT-141 activating melanocortin receptors that have gone quiet due to stress, aging, or hormonal changes. The signals radiating outward represent something patients describe as "feeling like I actually want to again."
In two Phase 3 trials with over 1,200 women, bremelanotide (PT-141) significantly increased sexual desire and reduced distress related to low desire compared to placebo. Approximately 50% of treated women reported meaningful improvements in desire, leading to FDA approval for hypoactive sexual desire disorder in premenopausal women.
In a randomized trial of 342 ED patients non-responsive to sildenafil, bremelanotide (PT-141) produced successful erectile response in 34% versus 9% on placebo (p=0.03), with significantly greater intercourse satisfaction. Unlike PDE5 inhibitors which act on peripheral vasculature, bremelanotide initiates erection via hypothalamic melanocortin receptors — a completely separate pathway that explains its efficacy in men who fail conventional ED medications. The FDA approved bremelanotide (Vyleesi) in 2019, validating the melanocortin approach.
The Transformation

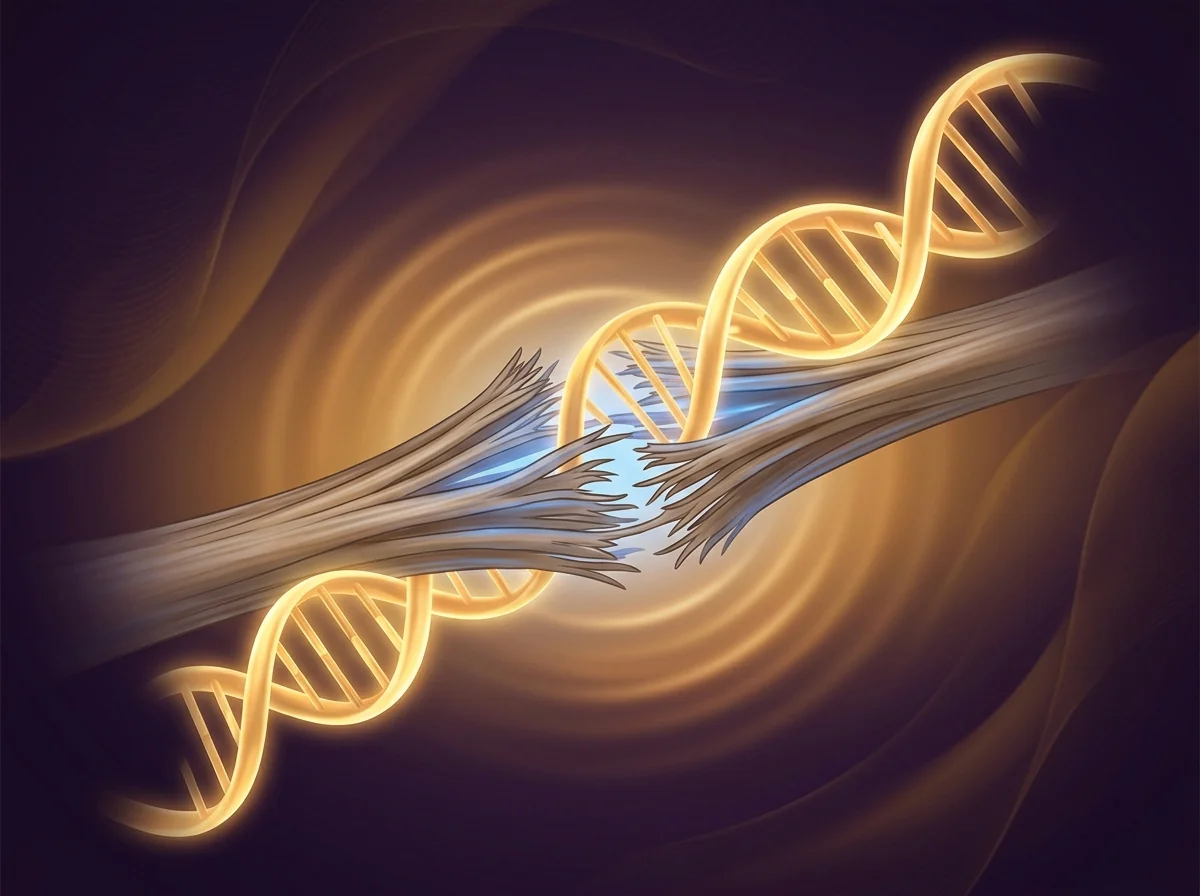
This comparison captures what patients experience in a way that's hard to put into words. On the left: dim, barely-connected neurons — this represents what's happening in the brain when desire fades. Many patients describe this as "I love my partner but I just don't feel anything anymore." The RECONNECT pivotal trials, published in the Journal of Sexual Medicine with over 1,200 women, found that approximately 50% reported meaningful improvements in desire — leading to FDA approval for hypoactive sexual desire disorder. On the right, after PT-141: the same neural network is alive, every connection is firing, the entire system is engaged. A follow-up study in Obstetrics & Gynecology confirmed a 38% increase in desire scores, with effects lasting up to 24 hours from a single dose.
FDA approval study demonstrated that PT-141 increased satisfying sexual events by 25% and desire scores by 38% in women with HSDD. Unlike hormonal treatments, PT-141 works within 30-60 minutes on an as-needed basis, with effects persisting up to 24 hours. The most common side effect was temporary mild nausea, which diminished with repeated use.
Mechanistic review established that MC3R and MC4R activation in the hypothalamic paraventricular nucleus generates pro-sexual signaling independent of androgen status. The authors explained why PT-141 produces erectile response in men who fail PDE5 inhibitors — the central pathway bypasses the peripheral vasodilation that PDE5 inhibitors target. Combined central-plus-peripheral protocols showed additive benefit.
52-week extension of the RECONNECT trials documented sustained improvements in desire, arousal, and sexual satisfaction without tachyphylaxis. Side effect profile (primarily mild transient nausea) improved over the treatment period as patients adapted. Discontinuation for efficacy loss was minimal — confirming durable central melanocortin-mediated response.
What to Expect
On-Demand Initiation
Provider-directed on-demand plan begins. First-use nausea can occur and is usually transient. Response pattern varies; most patients refine timing with provider guidance.
Responsive Phase
Consistent central desire and arousal response. Most patients report reliable response within 30-45 minutes with effects lasting 6-12 hours. Nausea typically diminishes after the first 3-5 doses.
Sustained Use
On-demand pattern becomes predictable. Long-term extension trials (52 weeks) showed no tachyphylaxis — the response pattern remains stable over time with appropriate dosing.
Your Protocol at a Glance
Ideal For
Men or women experiencing low libido, difficulty with arousal, or sexual dysfunction. Works regardless of the cause — hormonal, psychological, or age-related.
Safety & Considerations
- Provider supervision required with baseline cardiovascular assessment
- Most common side effect: transient nausea (1-2 hours) in 30-40% of patients with first doses; diminishes with repeated use
- Causes mild transient blood pressure elevation — contraindicated in uncontrolled hypertension, recent MI, or stroke
- Not recommended during pregnancy, lactation, or in patients with recent cardiovascular events
- Works for both men and women — FDA-approved for premenopausal women with HSDD; off-label but evidence-based for men with ED non-responsive to PDE5 inhibitors
Stacks Well With
Patients commonly combine PT-141 (Bremelanotide) with these for complementary or synergistic effects:
Frequently Asked Questions
Ready to Start PT-141 (Bremelanotide)?
Add this to your plan so we can confirm the right protocol, pricing, and next steps at your consultation. Or book directly.
Cancellation Policy: A $50 non-refundable deposit is required to confirm your appointment for all consultations — our front desk will reach out to collect it after you book. The deposit is applied toward your visit cost. We require at least 24 hours' notice for cancellations or changes. No-shows or late cancellations will forfeit the deposit. By booking, you agree to these terms.
Business hours: Mon–Thu 8am–6pm · Fri 8am–12pm