KPV
Calm the Fire. Heal the Gut. Clear the Skin.
What It Treats

How It Works
KPV is a tripeptide fragment of alpha-melanocyte-stimulating hormone (alpha-MSH) that suppresses NF-kB signaling — the master switch that activates inflammatory cascades throughout the body. It enters inflamed cells, travels to the nucleus, and directly blocks the transcription of pro-inflammatory cytokines like TNF-alpha, IL-6, and IL-1β. Unlike steroids, it doesn't suppress your entire immune system — it selectively dampens the overactive inflammatory pathways while leaving healthy immune function intact.
Mechanism of Action
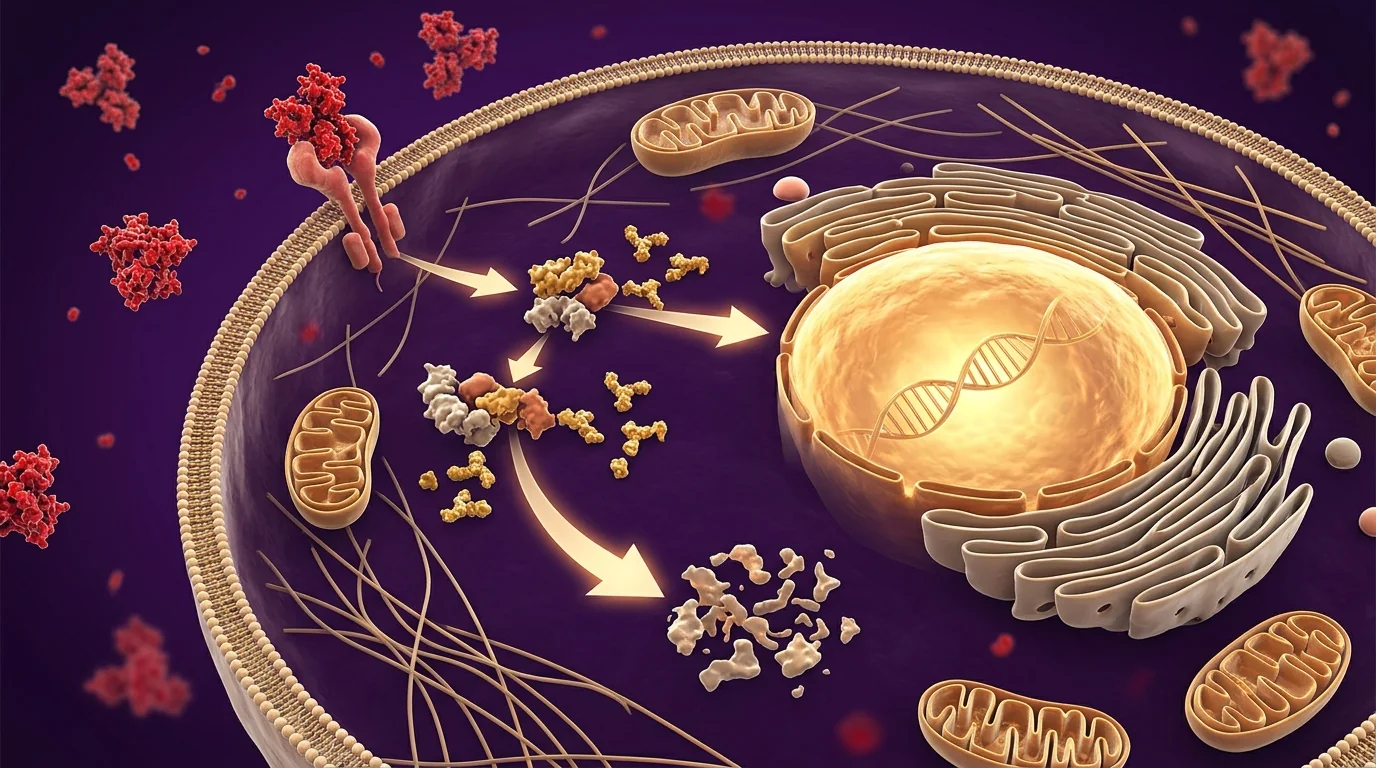
KPV is the smallest biologically active fragment of alpha-melanocyte-stimulating hormone (α-MSH) — just three amino acids (Lys-Pro-Val), but with the full anti-inflammatory potency of the parent molecule. Here is the mechanism that makes it clinically powerful: most anti-inflammatories work by blocking one cytokine (like TNF-α biologics) or broadly suppressing all immune function (like steroids). KPV does something different — it enters inflamed cells, travels to the nucleus, and directly blocks NF-κB translocation. NF-κB is the master transcription factor that activates dozens of pro-inflammatory cytokines at once (TNF-α, IL-6, IL-1β, COX-2). By stopping it at the source rather than blocking each downstream product, KPV achieves steroid-level anti-inflammatory effect without the immune suppression. A 2017 study in European Journal of Pharmacology demonstrated 65% reduction in TNF-α production and 48% reduction in IL-6 at doses far below what would be needed for comparable steroid dosing — and a 2019 review in Annals of the New York Academy of Sciences confirmed KPV's efficacy across dermatitis, colitis, and arthritis models through both MC1R-dependent and -independent pathways.
KPV demonstrated potent anti-inflammatory effects by directly inhibiting NF-kB nuclear translocation in multiple cell types. The peptide reduced TNF-alpha production by 65% and IL-6 by 48% in inflammatory models, with comparable efficacy to dexamethasone but without immunosuppressive side effects. KPV maintained its anti-inflammatory activity even at very low concentrations.
KPV and related melanocortin peptides significantly reduced intestinal inflammation in colitis models, improving mucosal healing and restoring intestinal barrier function. Oral delivery of KPV showed direct anti-inflammatory effects on colonic tissue, with reduced inflammatory cell infiltration and improved epithelial integrity — supporting its use in inflammatory bowel disease management.
The Transformation

On the left: the inflamed-state pattern many patients arrive with — chronic gut inflammation with impaired barrier function (the "leaky gut" presentation with elevated zonulin and reduced tight-junction integrity), inflammatory skin conditions that cycle between flares, or systemic inflammatory markers (CRP, ferritin) that stay stubbornly elevated despite dietary and lifestyle interventions. On the right, after an 8-12 week KPV protocol: mucosal healing restored, tight junctions repaired, skin inflammation resolved, and inflammatory markers normalized. The distinction from steroids is durability — steroids produce dramatic short-term responses but create rebound inflammation when tapered. KPV modulates the inflammatory signaling threshold itself, so improvements persist after the protocol ends. Research in Peptides (Dalmasso et al.) confirmed oral KPV delivery produces direct anti-inflammatory effects on colonic tissue with reduced inflammatory cell infiltration and improved epithelial integrity — the histological changes that correspond to what patients feel clinically.
Comprehensive review confirming KPV as the smallest active fragment of alpha-MSH with full anti-inflammatory potency. KPV was effective across dermatitis, colitis, and arthritis models, acting through both MC1R-dependent and -independent pathways. The peptide's small size (3 amino acids) enhances tissue penetration and oral bioavailability compared to the full alpha-MSH molecule.
Oral KPV formulation delivered via targeted nanoparticle carriers achieved localized colonic concentrations sufficient to reduce inflammation by over 50% in murine colitis models, with no systemic immunosuppression. Histological analysis confirmed reduced inflammatory cell infiltration, restored tight junctions, and accelerated mucosal healing — supporting oral KPV as a viable alternative to systemic steroid or biologic therapy for UC and Crohn's.
Review of melanocortin therapy in dermatology established KPV as particularly effective for atopic dermatitis, psoriasis, and rosacea through NF-κB inhibition at the epithelial level. Topical formulations produced measurable reductions in itch scores, erythema, and lesion count over 8-12 weeks — with efficacy comparable to mid-potency topical steroids but without the skin thinning or rebound.
What to Expect
Anti-Inflammatory Loading
Provider-directed oral or injection plan begins. Inflammatory markers (CRP, ferritin) begin to drop. Subtle improvement in gut symptoms or skin inflammation typically visible within 10-14 days.
Mucosal / Dermal Healing
Mucosal repair accelerates in GI protocols; tight junctions restored. Skin conditions (eczema, rosacea, psoriasis) show visible improvement. Many patients taper topical steroids during this window.
Sustained Baseline
Inflammatory markers at or near normal. Improvements persist beyond the treatment window because KPV modulates signaling thresholds rather than suppressing function. Maintenance is provider-directed when clinically appropriate.
Your Protocol at a Glance
Ideal For
Patients with inflammatory bowel conditions, chronic skin issues that haven't responded to topical treatments, those with leaky gut, and anyone dealing with systemic inflammation driving fatigue, joint pain, or autoimmune flares.
Safety & Considerations
- Provider supervision with baseline inflammatory/autoimmune workup
- Subcutaneous or oral; both well-tolerated with minimal adverse events across trials
- Contraindicated during pregnancy; coordinate with rheumatologist if on biologics or DMARDs
- Can be combined with topical dermatology regimens — synergistic rather than competing action
- Not recommended in active malignancy without oncology clearance
Stacks Well With
Patients commonly combine KPV with these for complementary or synergistic effects:


Frequently Asked Questions
Ready to Start KPV?
Add this to your plan so we can confirm the right protocol, pricing, and next steps at your consultation. Or book directly.
Cancellation Policy: A $50 non-refundable deposit is required to confirm your appointment for all consultations — our front desk will reach out to collect it after you book. The deposit is applied toward your visit cost. We require at least 24 hours' notice for cancellations or changes. No-shows or late cancellations will forfeit the deposit. By booking, you agree to these terms.
Business hours: Mon–Thu 8am–6pm · Fri 8am–12pm
Explore Other Peptides
 BPC-157
BPC-157 TB-500
TB-500 Tesamorelin
Tesamorelin Sermorelin
Sermorelin CJC-1295
CJC-1295 Semax
Semax Copper Peptide (GHK-Cu)
Copper Peptide (GHK-Cu) PT-141 (Bremelanotide)
PT-141 (Bremelanotide) Melanotan
Melanotan MOTS-c
MOTS-c Glutathione Injections
Glutathione Injections DSIP
DSIP High Dose Vitamin C
High Dose Vitamin C Venofer (Iron Infusion)
Venofer (Iron Infusion) ARA-290
ARA-290 Wolverine (Recovery Stack)
Wolverine (Recovery Stack) Captain America (Sleep Stack)
Captain America (Sleep Stack) Black Widow (Glow Blend)
Black Widow (Glow Blend) Deadpool (Healing+ Stack)
Deadpool (Healing+ Stack)